Panintorn Thunwiriya, Tanaporn Phetruen, Phaewa Chaiwijit, Chaiyawat Tonawannakorn, Kanokwan Dekham, Nalumon Thadtapong, Soraya Chaturongakul, Varodom Charoensawan , Sittinan Chanarat, Danaya Pakotiprapha, Prut Hanutsaha
Abstract
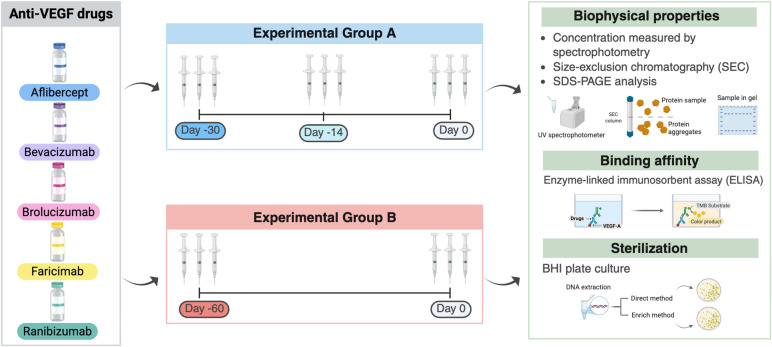
Intravitreal injection anti-vascular endothelial growth factor (VEGF) drugs offer significant benefits for individuals with retinal diseases, yet high cost and limited availability impede widespread use. Compounding and repackaging present a potential strategy for better drug utilization; however, earlier studies addressing the efficacy and sterility of repackaged anti-VEGF drugs focused on individual or few drugs, limiting their ability to provide comparative insights across different drugs and experimental settings. In this study, we survey the stability, efficacy, and sterility of five intravitreal anti-VEGF drugs, namely aflibercept, bevacizumab, brolucizumab, faricimab, and ranibizumab, after repackaging and storage for 14, 30, and 60 days. The drugs were aseptically repackaged into a minimum of three syringes per time points, which were all examined using one consistent protocol comprising six different experimental techniques: spectrophotometry, sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE), size exclusion chromatography (SEC), enzyme-linked immunosorbent assay (ELISA), and microbial plating. All five intravitreal anti-VEGF drugs can be repackaged and stored in Tuberculin-type syringes up to 60 days, while maintaining sterility up to 60 days with minimal changes in protein concentrations, degradation products, aggregation, and functional binding to VEGF-A, if appropriate aseptic protocols are followed. Specific differences observed in biophysical properties between drugs were noted and discussed.
Supplementary Information: The online version contains supplementary material available at 10.1038/s41598-026-39102-5.
Keywords: Compounding pharmacy; Intravitreal anti-VEGF; Repackaging drugs; Storage times.

